
In conclusion Electron affinity is more difficult to analyse in molecules as their structure is shows complications. Electron affinity is the enthalpy change for the process: X((g))+erarrX((g))- The incoming electrons will experience a force of attraction with the effective charge of the nucleus. Negative electron affinity driven broadband absorption of Cs3+nPbnSb2I9+3n/GaN van der Waals heterostructures Department of Physics, National Laboratory of. Electron affinities are usually negative values, since energy is usually released (an exothermic energy change) when an electron is added to a neutral atom. The elements Chlorine strongly attracts electrons while mercury is the element with atoms that most weakly attract an electron. Electron affinity can be negative if the process releases energy. Non-metals typically have higher value than metals. If value is positive the process is exothermic and occurs spontaneously.Įach of these elements has a completely filled valence electron shell and an electron affinity that approaches zero. That is, the electron affinity is equal to the energy difference between the enthalpy of formation of a neutral species and the enthalpy of formation of the negative ion of the same structure. 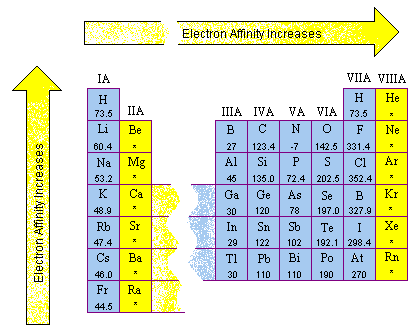
The value may be either positive or negative value, the negative value means energy must be present in order to attach an electron to the ion, here the capture of electrons is an endothermic process. The electron affinity, EA, of a molecule is, for negative ions or anions, the quantity that is analogous to the ionization energy for positive ions. It increases when it moves down a column or group and also shows an increase when it moves from left to right across a period or row( except for the noble gases). The diamond (100) surface was found to exhibit a negative electron affinity (NEA) following a 900 C anneal in ultrahigh vacuum. 
Electron affinity follows a particular path on the periodic table.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
